The operating layer forthe Pharma industry
Coincidence Labs helps biopharma teams turn fragmented systems and manual workflows into AI systems that constantly improve
Trusted by






Tech-driven innovation, people-driven change across the pharma value chain
Integrated withyour stack
We structure data from every system your teams use and digitize what's still on paper. From scattered silos to one unified layer that every AI agent can read, query, and act on.
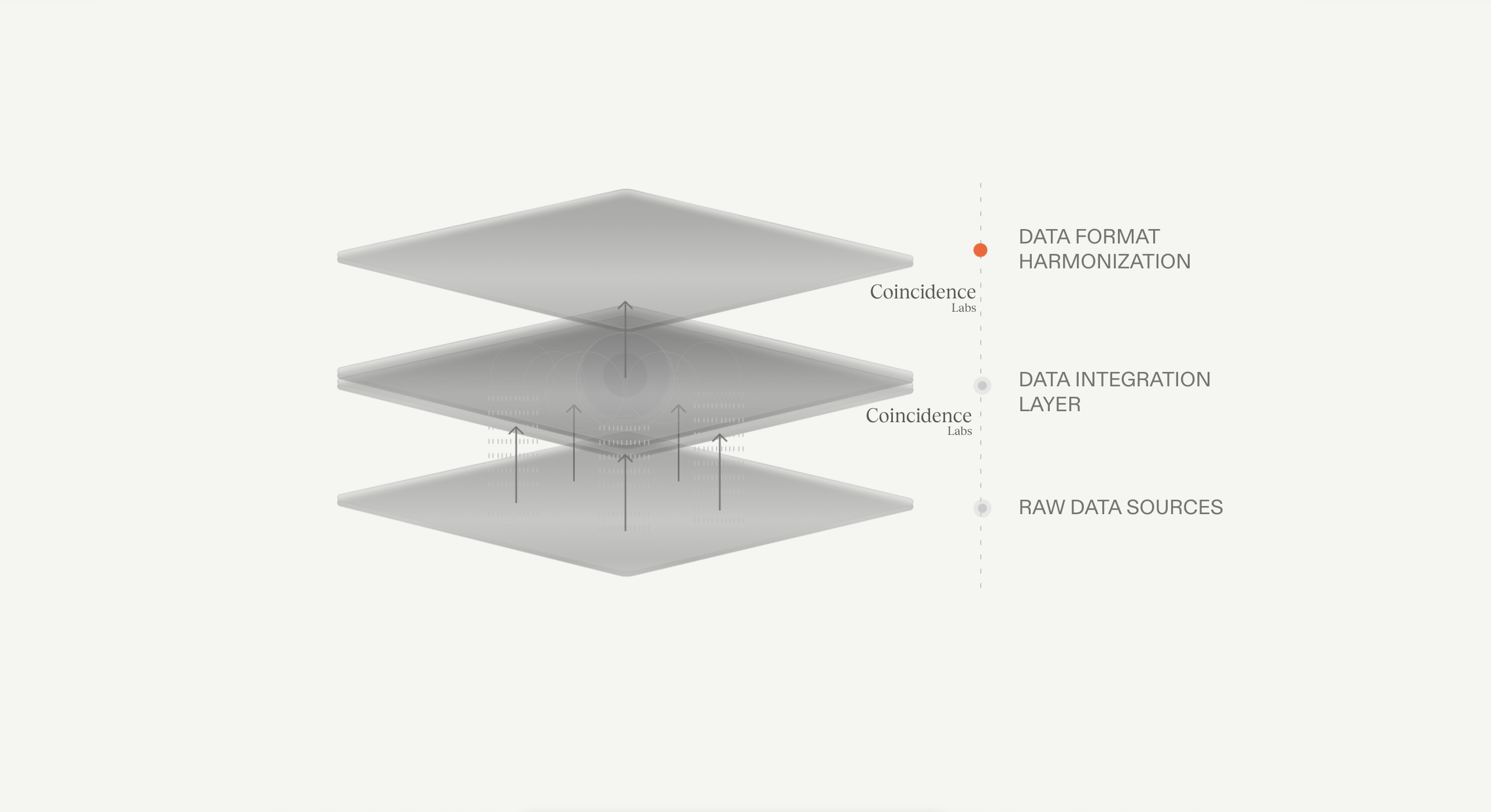
To turn manual processesinto repeatable AI systems
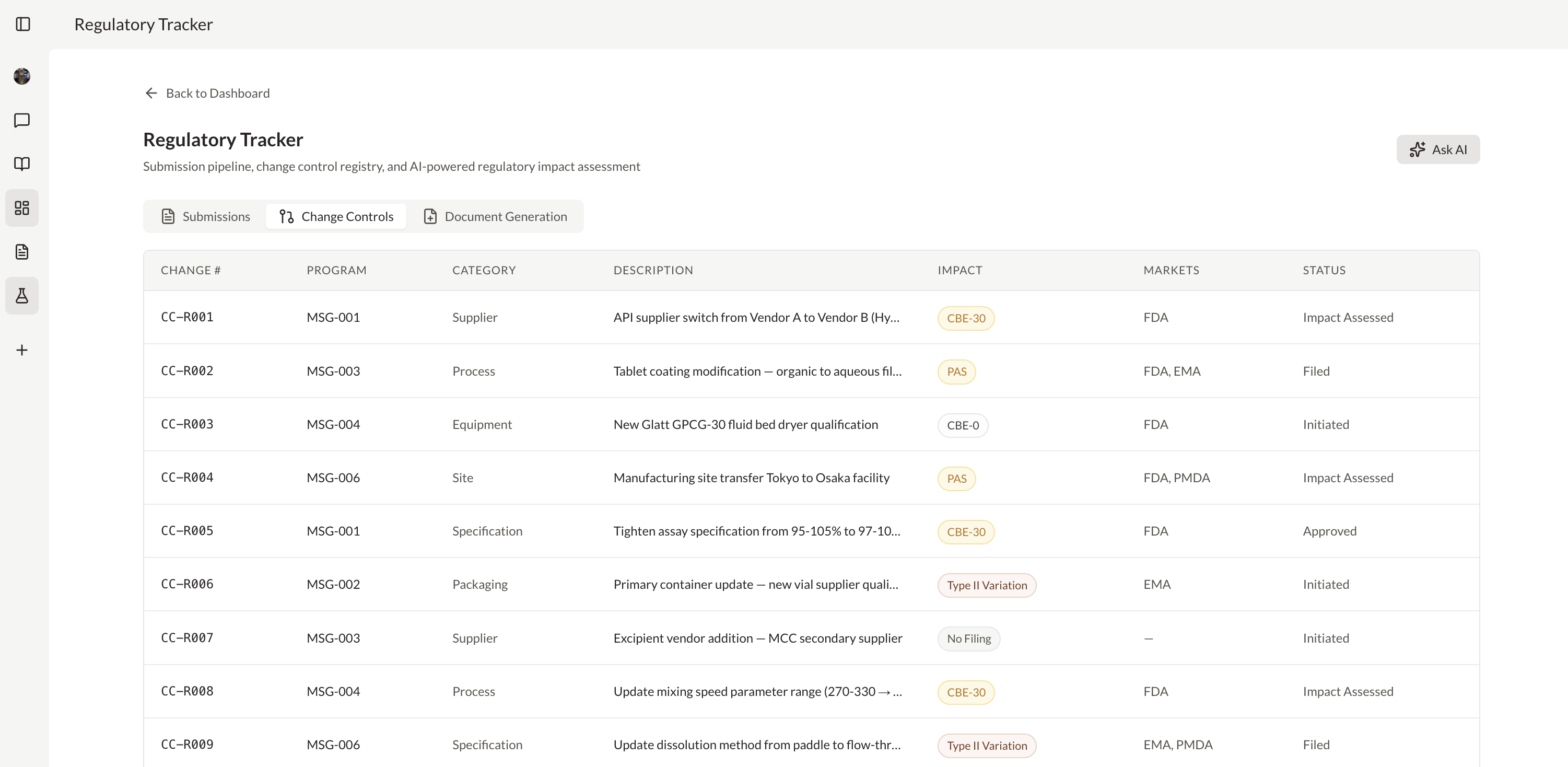
Complex, time-sensitive tasks like regulatory documentation don't need to be manual. AI agents execute them with deterministic logic and route to your experts when the decision requires it.
Workflow templates
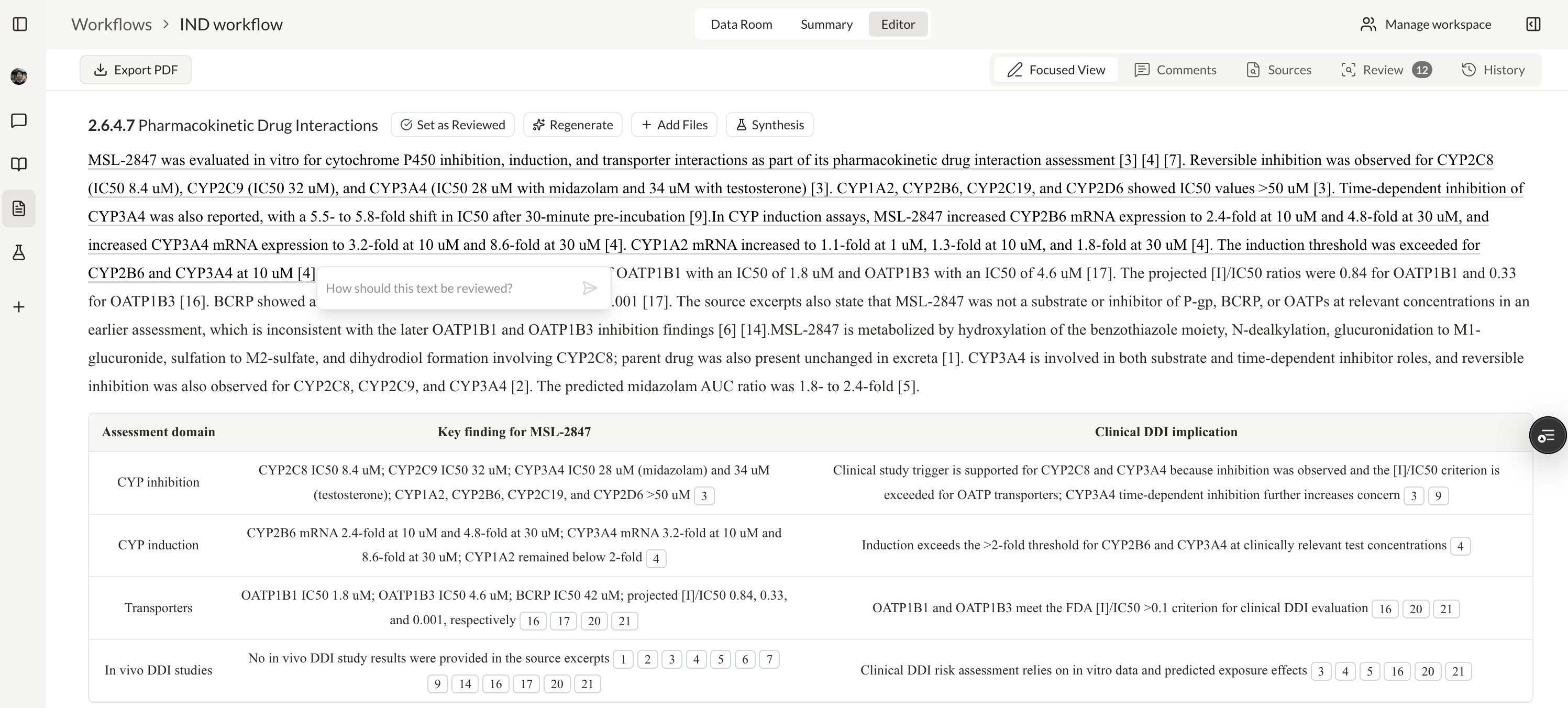
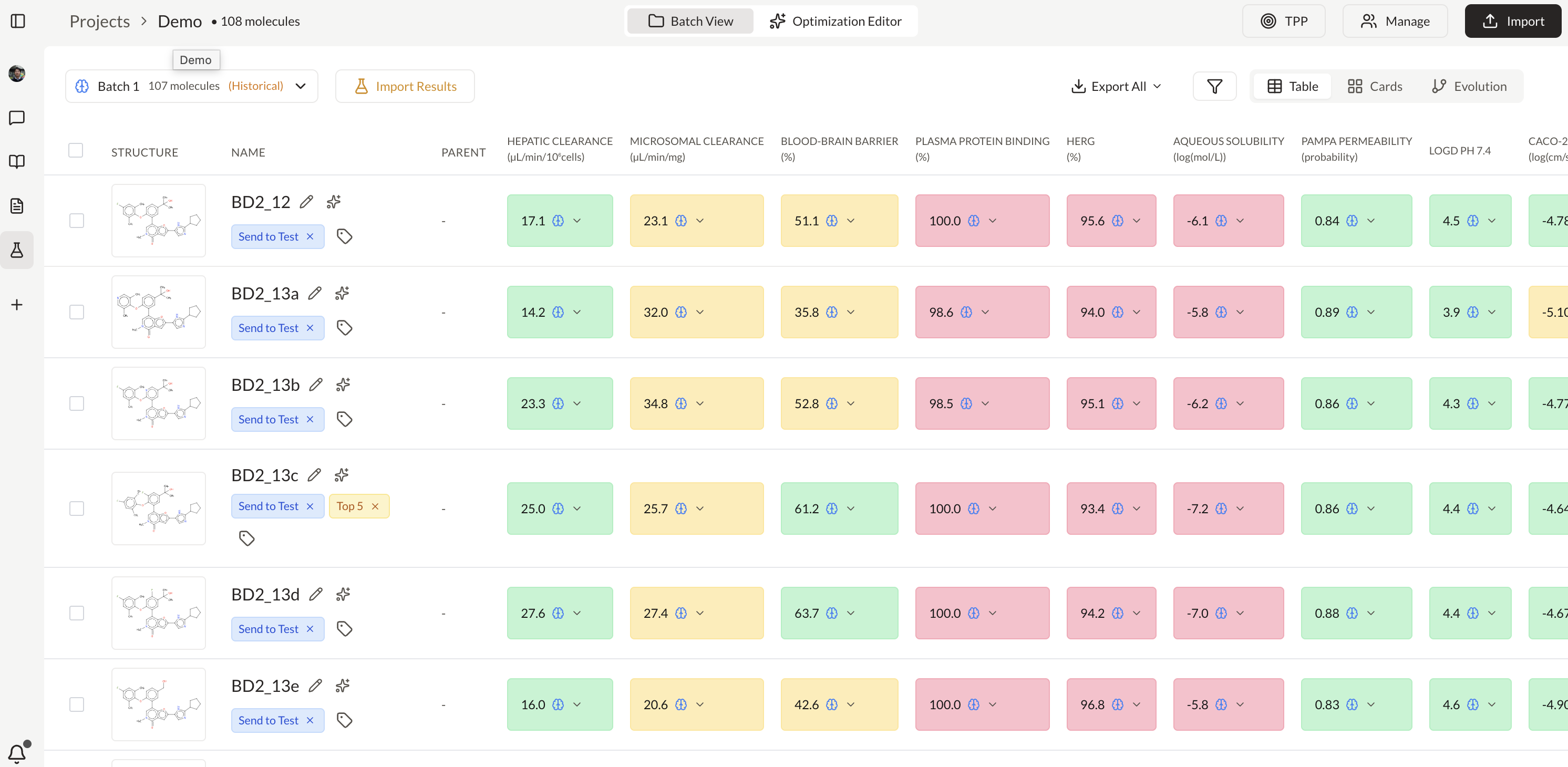
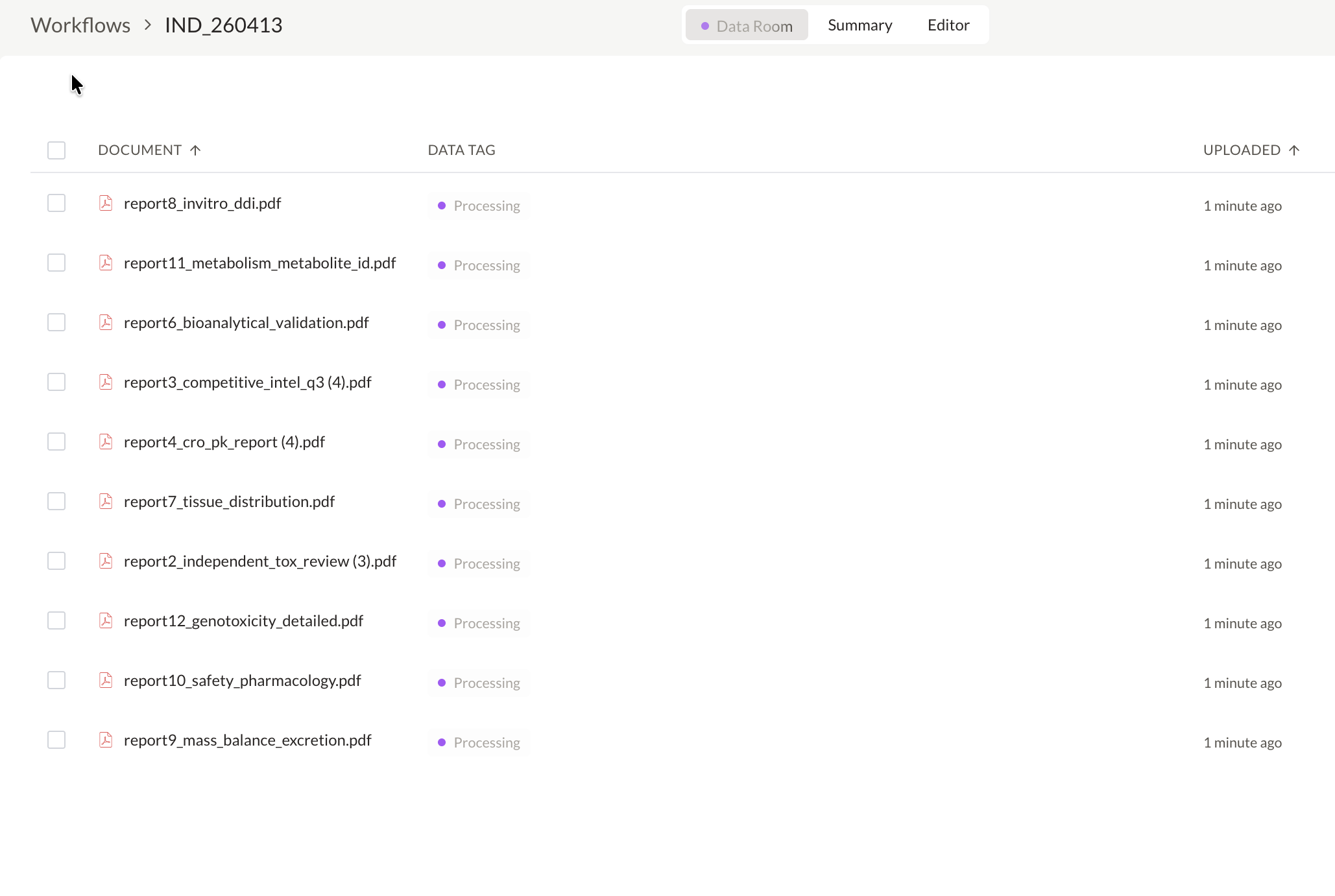
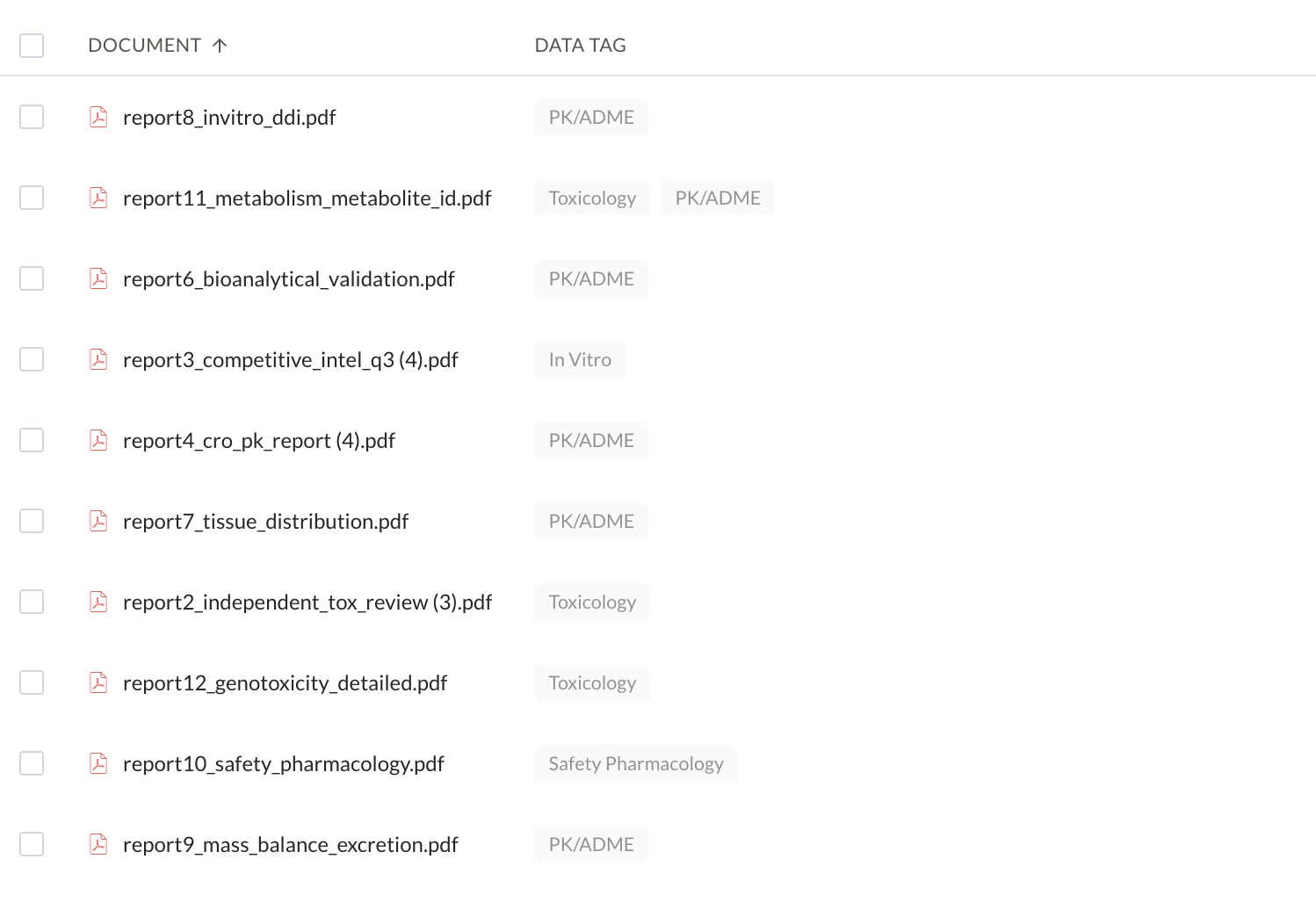
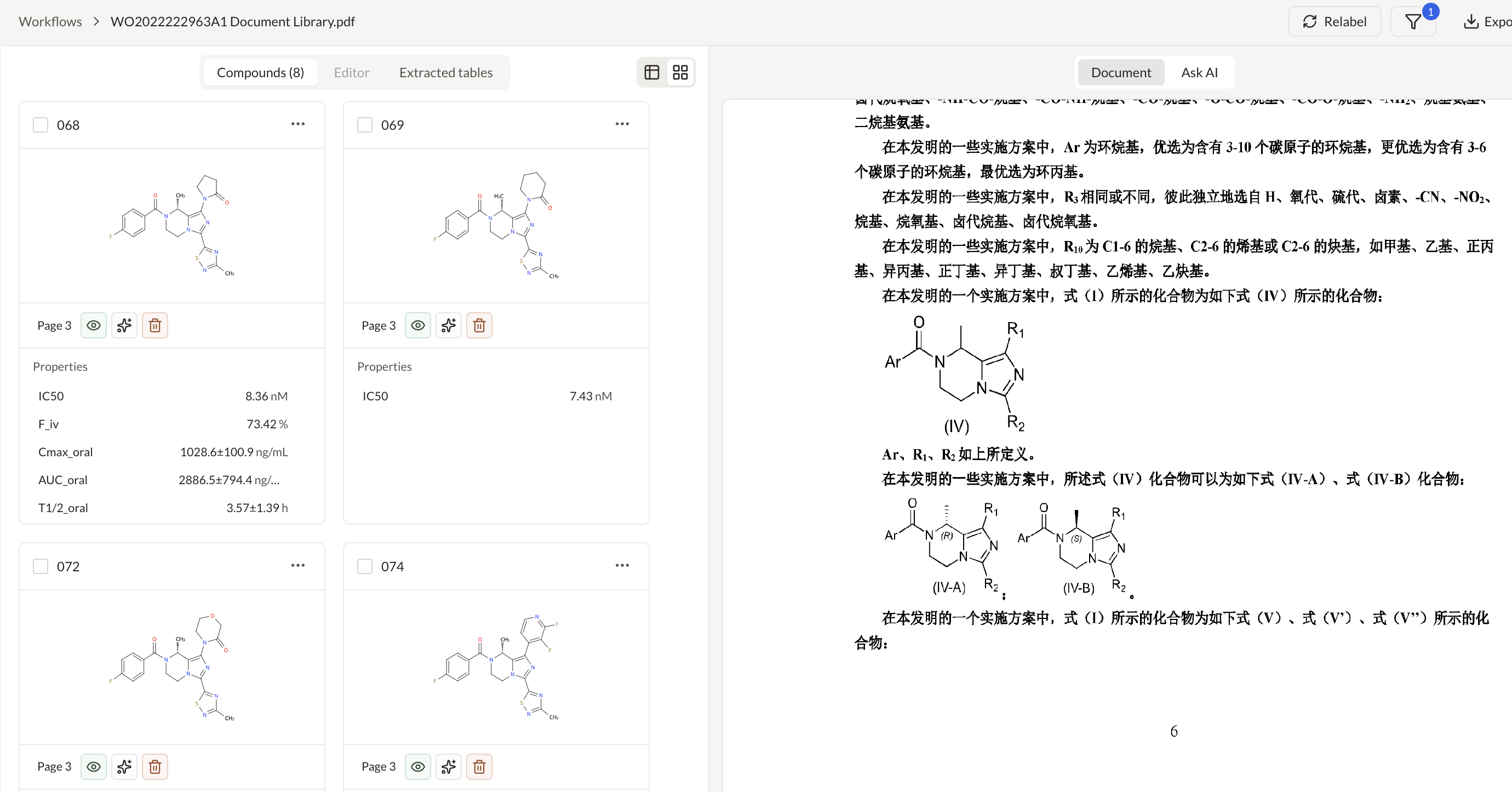
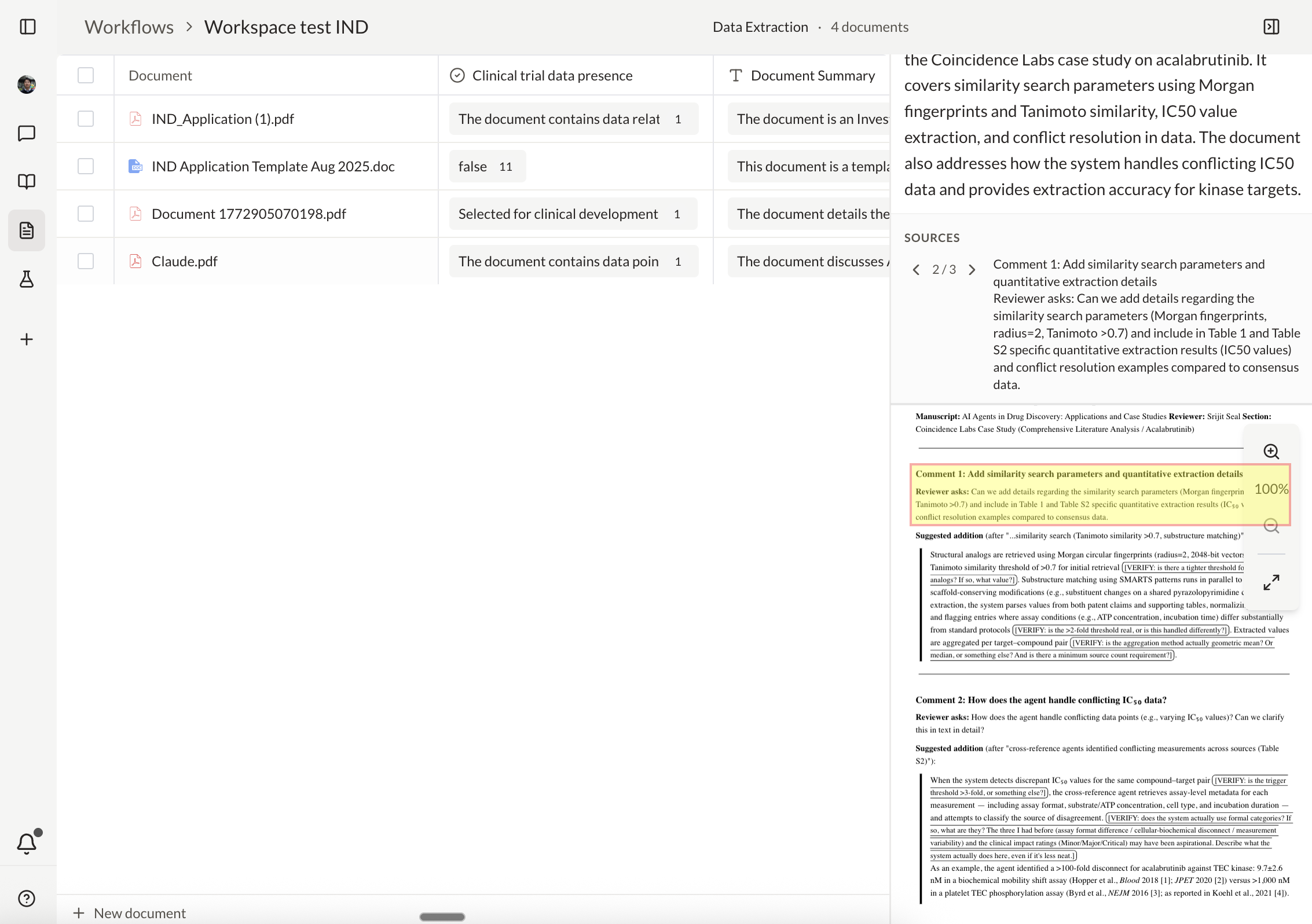
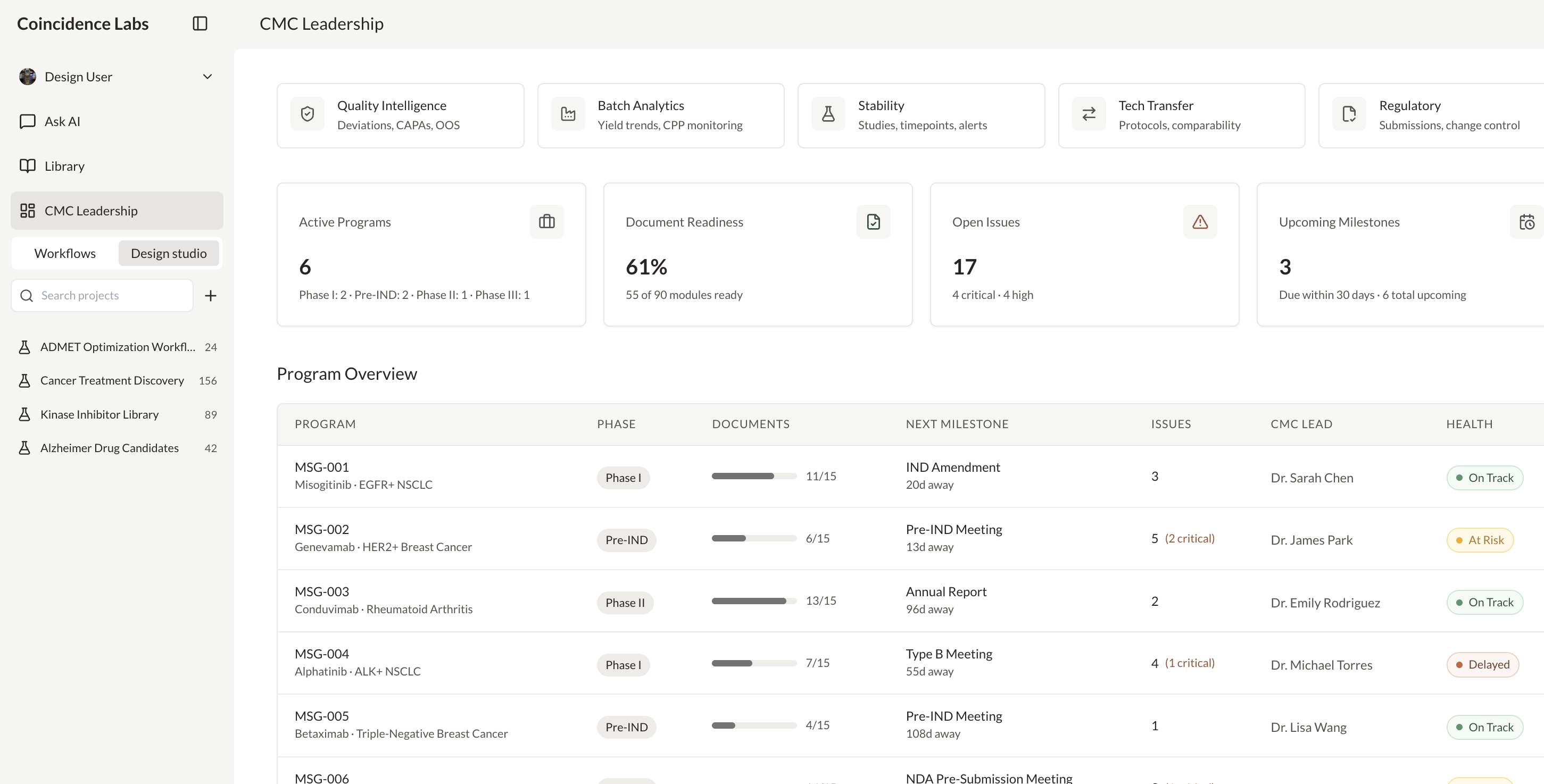
Create and manage IND application workspaces
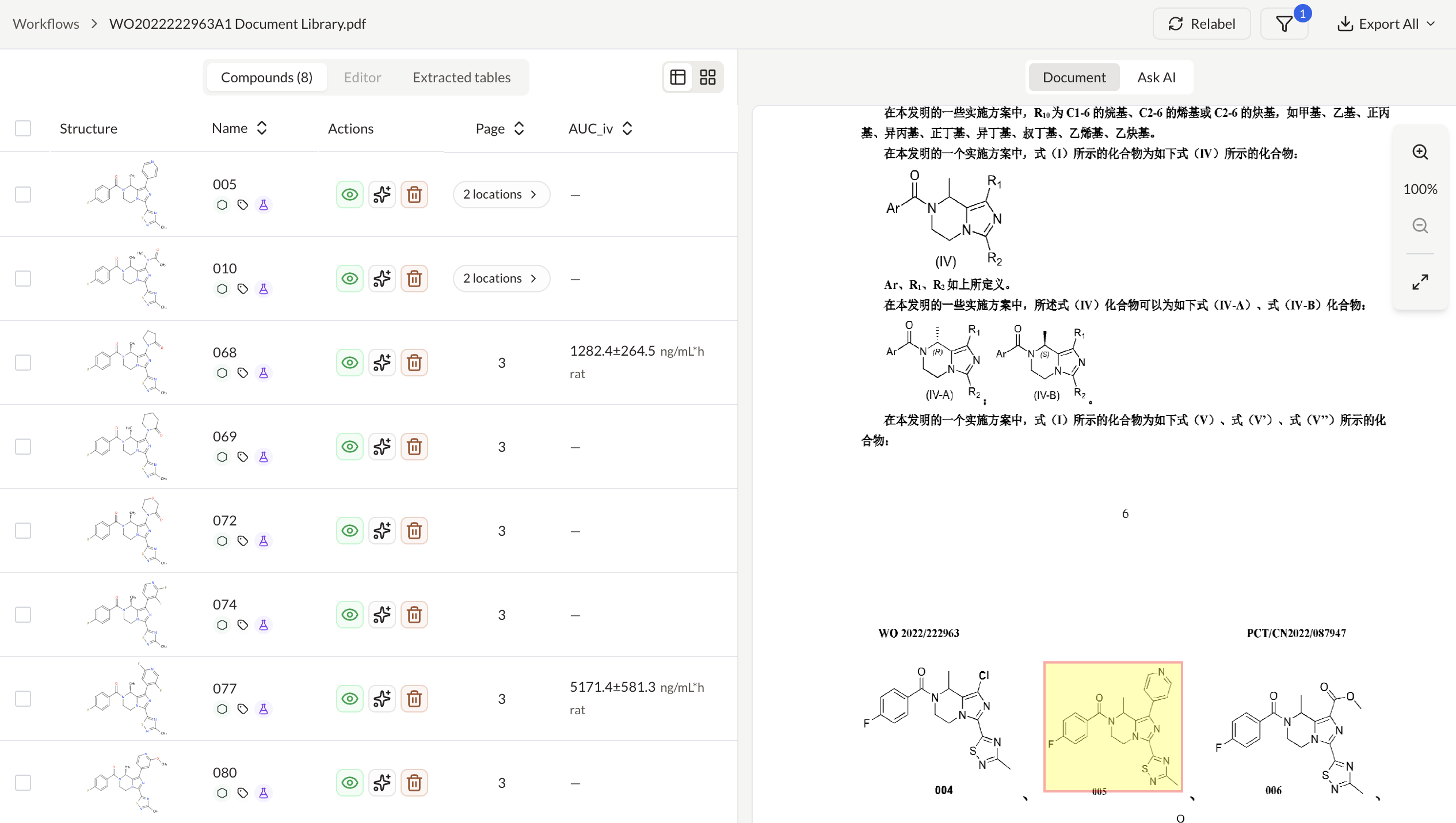
Analyze compounds with patents and clinical trials insights
Extract and structure claims from patent documents
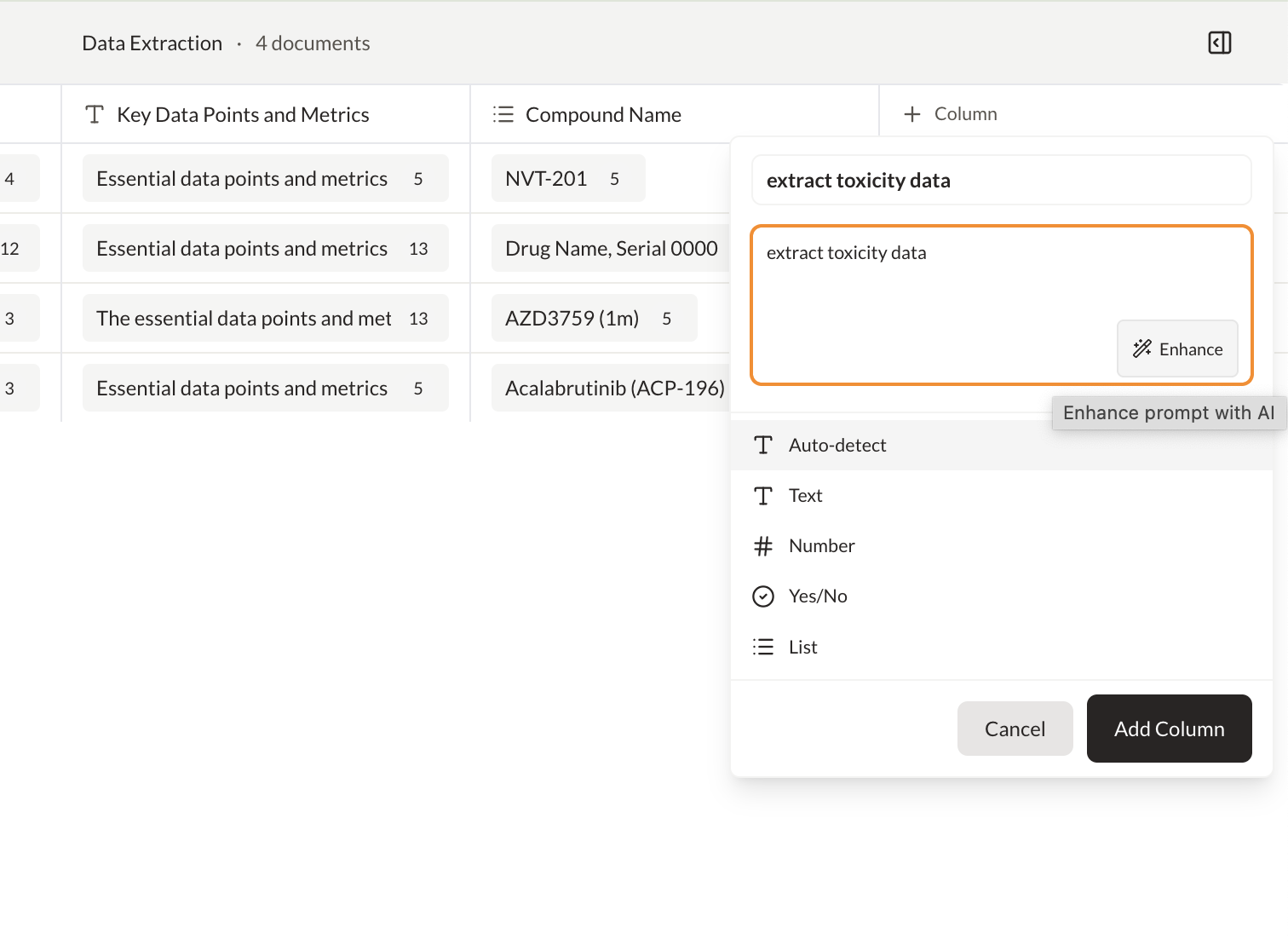
Ask questions across PDFs and map answers to a custom schema
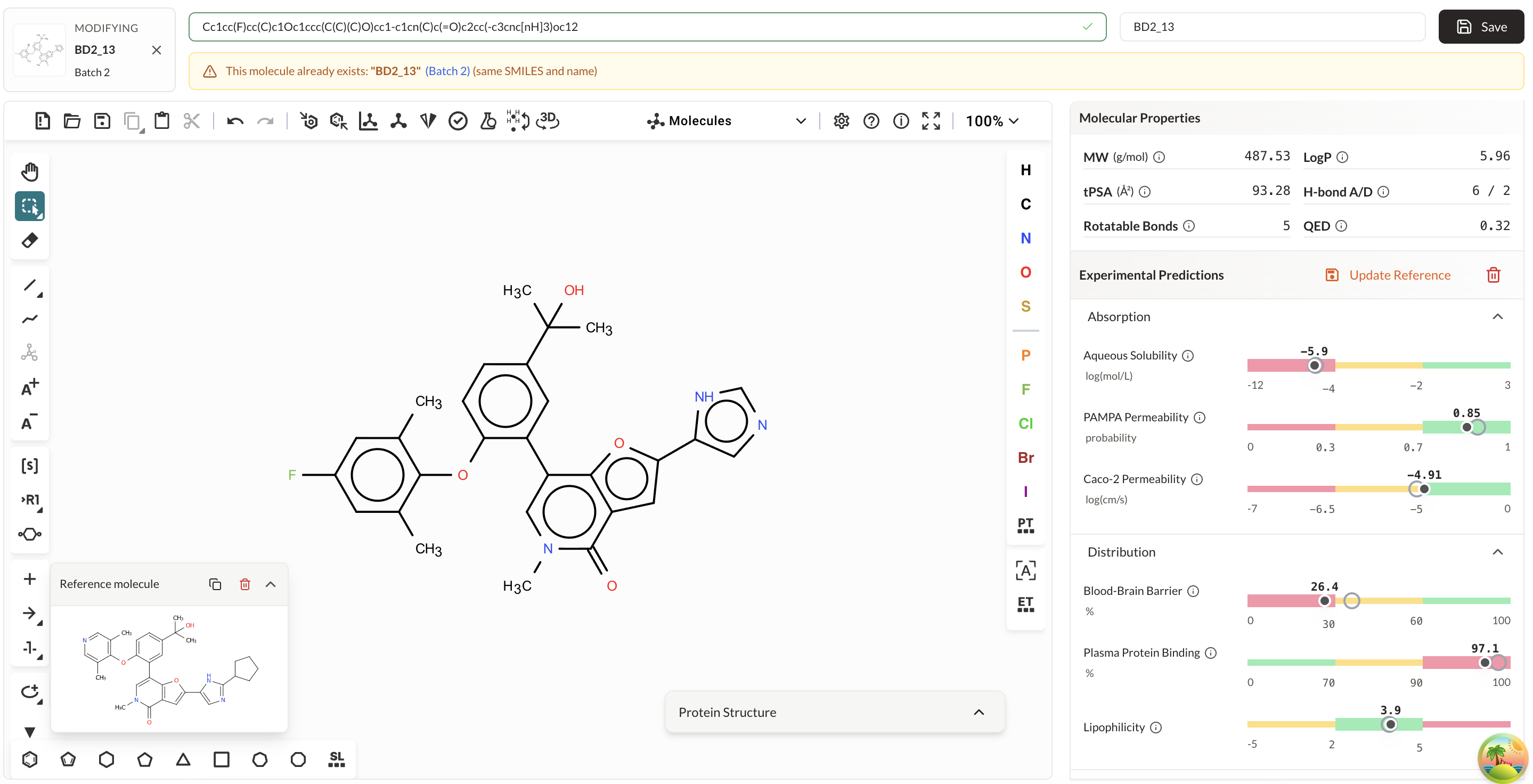
Test hypotheses against predictive models and optimize candidates
Classify, label, extract, and route CSRs, patents, and internal reports
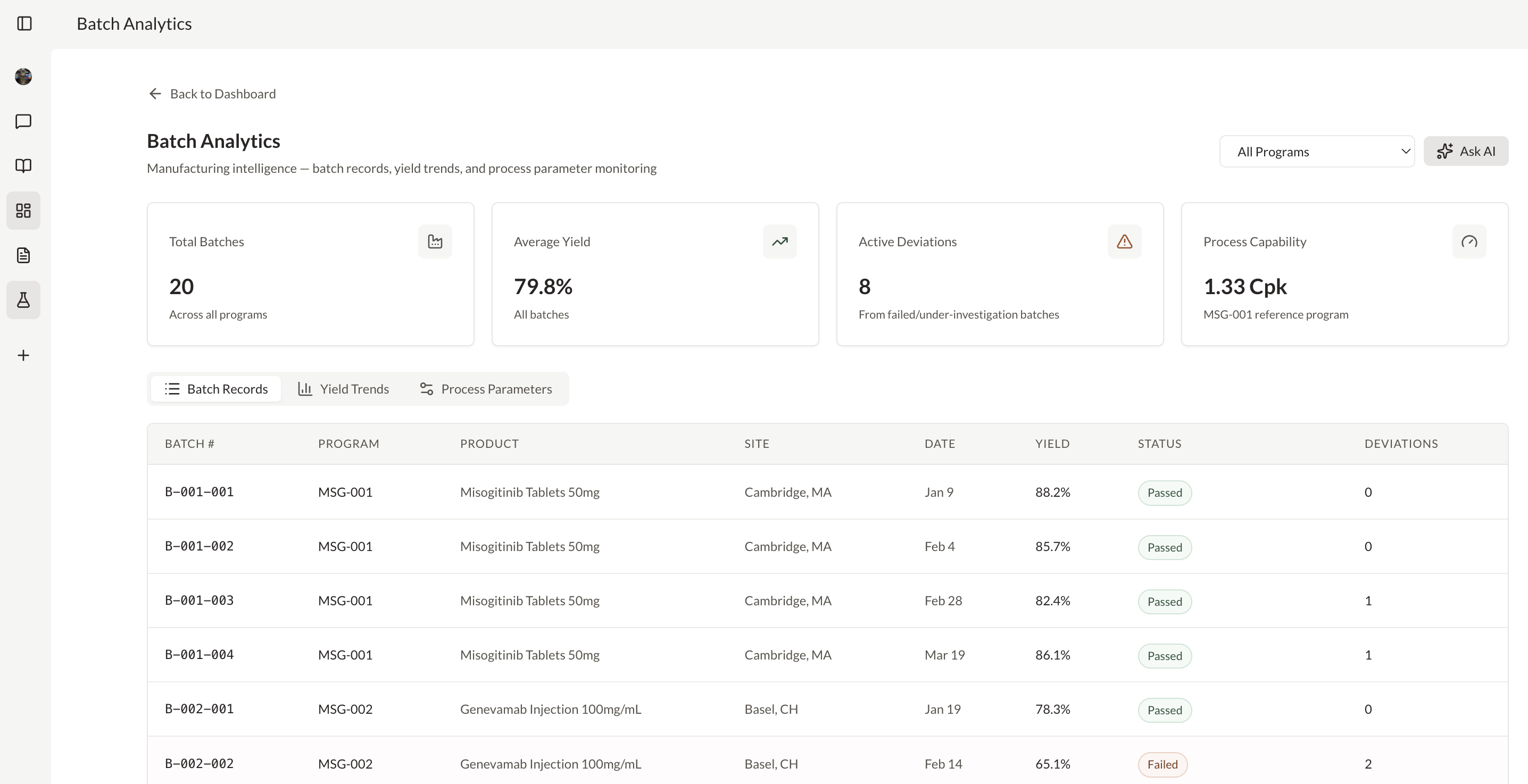
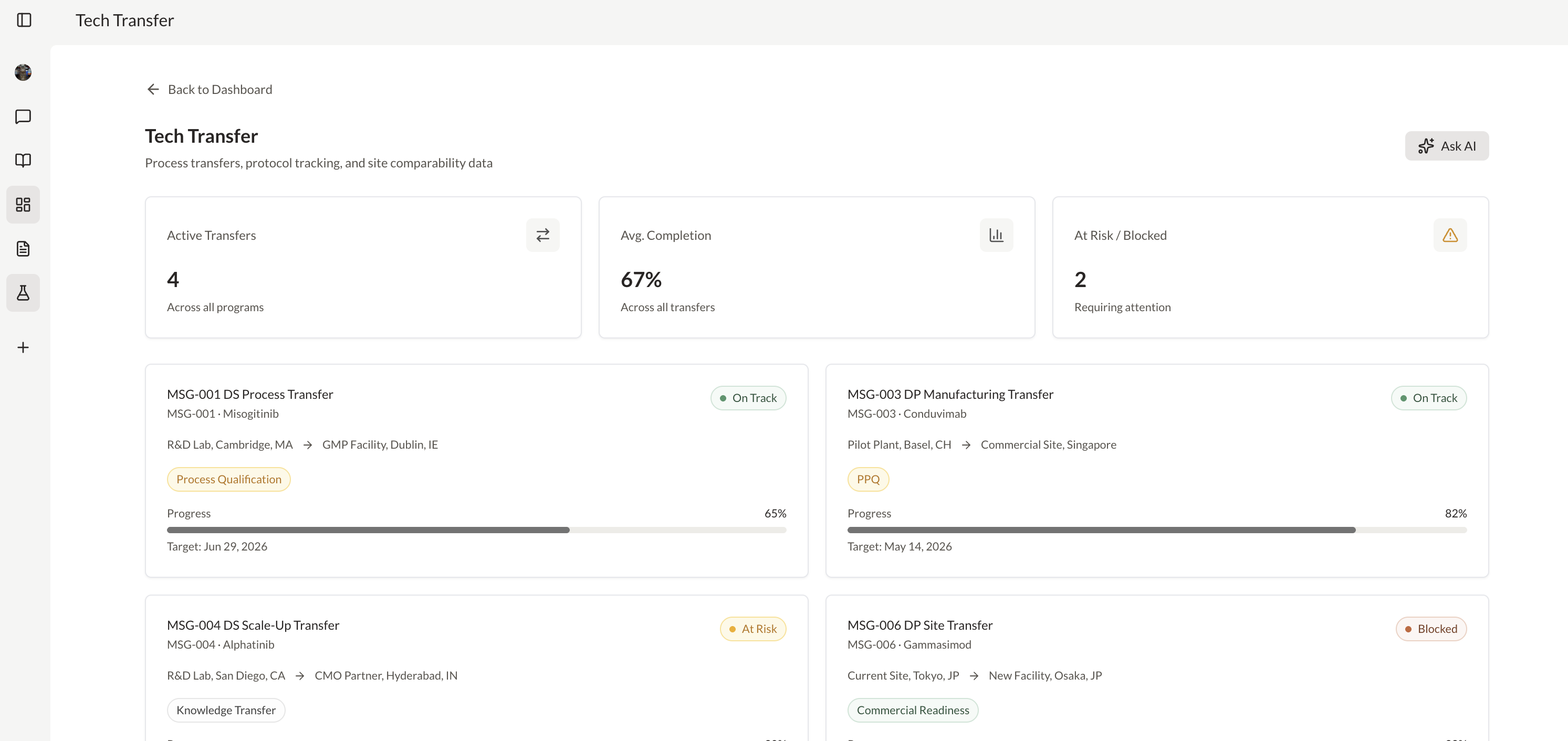
Generate real-time analytics across CMC, regulatory, or clinical data
Connect every document, decision, and dataset into one searchable layer
With on-demandintelligence
Your scientists, regulatory leads, and program managers each hold context no one else can access. Now your team can query it, trace decisions, and act on it across functions, in real time.
Built for regulated environmentsby design
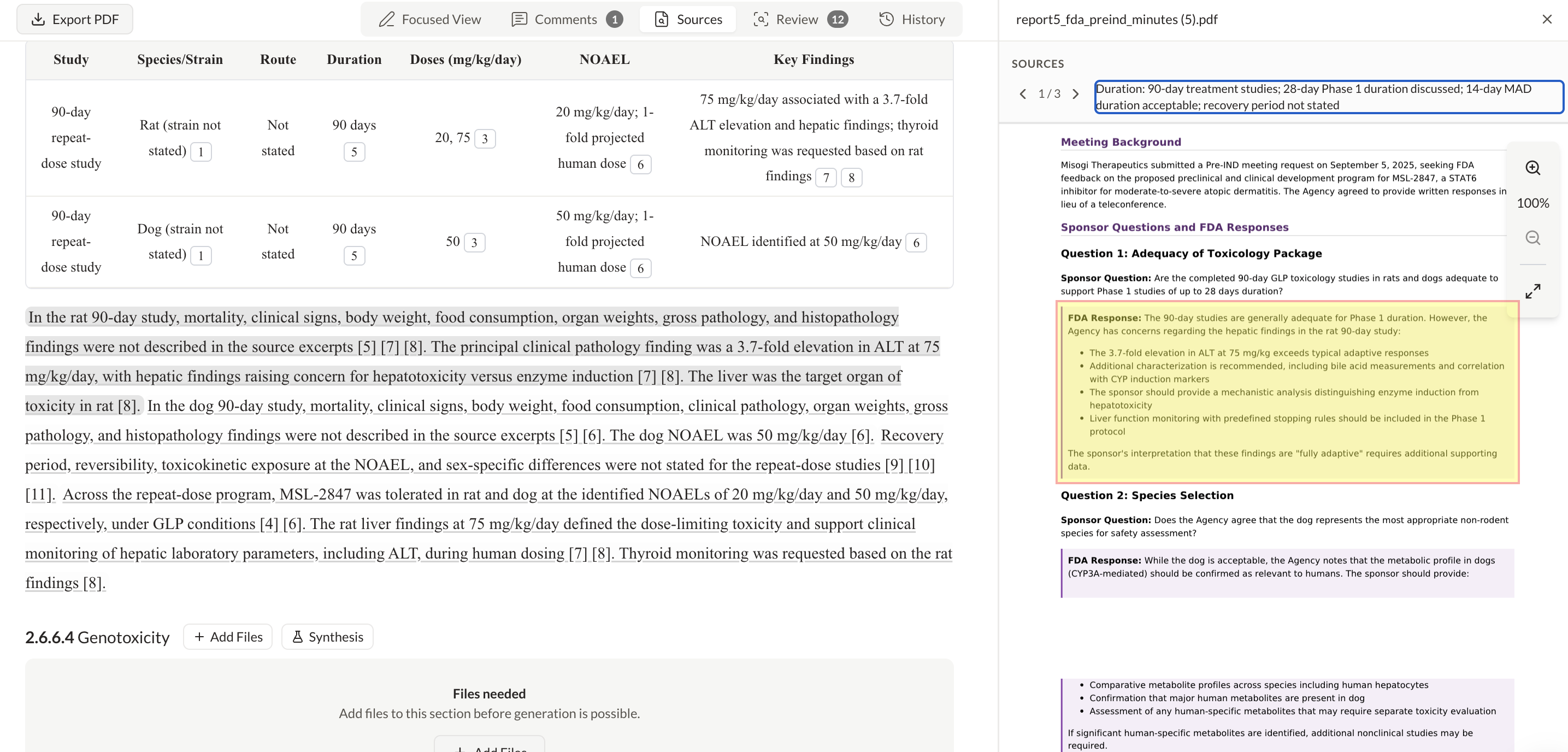
Every agent action is visible and traceable. Your team tracks decisions across functions in real time, and agents escalate to the right expert when review is required.
Audit Trail
LiveVerified protocol compliance against ICH E6(R3)
09:41:12Flagged inconsistency in inclusion criteria across v2.1 and v3.0
09:41:18Escalated finding for protocol amendment review
09:41:23Reviewed amendment — classified as non-substantial
14:23:08Logged approval — ref. RA-2024-0847
14:23:14Not another tool. The AI system that scales across teams and projects.

Use cases
Designed by industry experts
How we work
A clear path to working
AI systems and outcomes
Real transformation is built aligning technology and people to your internal processes. Our engineers work alongside your team as embedded partners to turn high-potential use cases into working AI systems that run across your organization.
AI Workshop
We work with your team to map internal processes, identify the highest-impact opportunities for AI, and the AI Champions who will drive adoption from within.
Scope
We define the business case and build a lightweight proof of concept to validate feasibility and value.
Pilot
We roll out the first selected use cases in your environment and measure real operational impact.
Scale
Expand across teams and operations, compounding on the proven value of AI systems and the change driven by the internal champions behind true innovation.
AI Workshop
We work with your team to map internal processes, identify the highest-impact opportunities for AI, and the AI Champions who will drive adoption from within.
Scope
We define the business case and build a lightweight proof of concept to validate feasibility and value.
Pilot
We roll out the first selected use cases in your environment and measure real operational impact.
Scale
Expand across teams and operations, compounding on the proven value of AI systems and the change driven by the internal champions behind true innovation.
Enterprise-grade data protection.Encrypted, isolated, and compliant.



Enterprise security
All data is encrypted in transit using TLS 1.2 or higher, and at rest with AES-256 encryption. SOC 2 Type II and ISO 27001 controls.
Data sovereignty
Your data remains fully owned by you. Deploy single-tenant on-premise, on the cloud of your choice, or multi-tenant fully isolated.
Model transparency
Your data is never used to train our models or third-party models. Access to leading LLMs including GPT, Claude, and Gemini, with data residency options in the EU or US.
Access control
Granular user roles and permissions across teams and projects for secure collaboration.
Questions & Answers
Interested in seeing it in actionfor your team? Let's talk.
The operating layer for the Pharma industry